
§5 Voltammetry §5-1 Basic principle of polarograpy Voltammetry A group of analytical methods based on determining current flow – voltage curve during electrolysis Several types of methods based on Electrode type How the potential is applied How the current is measured
§5 Voltammetry §5-1 Basic principle of polarograpy Voltammetry A group of analytical methods based on determining current flow – voltage curve during electrolysis Several types of methods based on Electrode type How the potential is applied How the current is measured
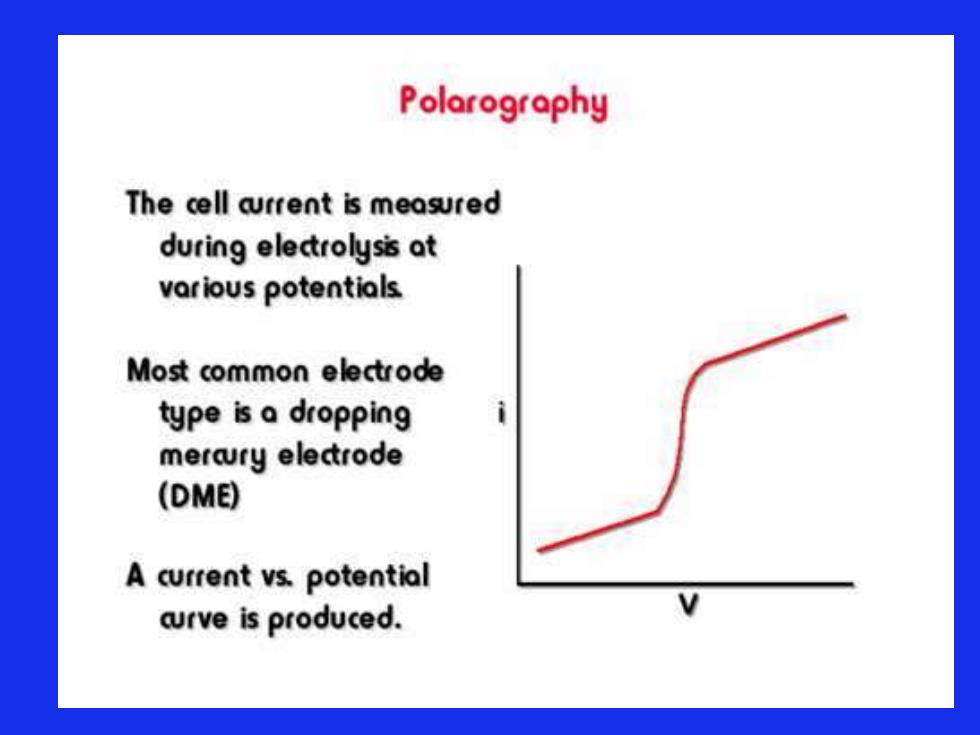
Polarography The cell arrent is measured during electrolysis at various potentials Most common electrode type is a dropping mercury electrode (DME) A current vs.potential aurve is produced
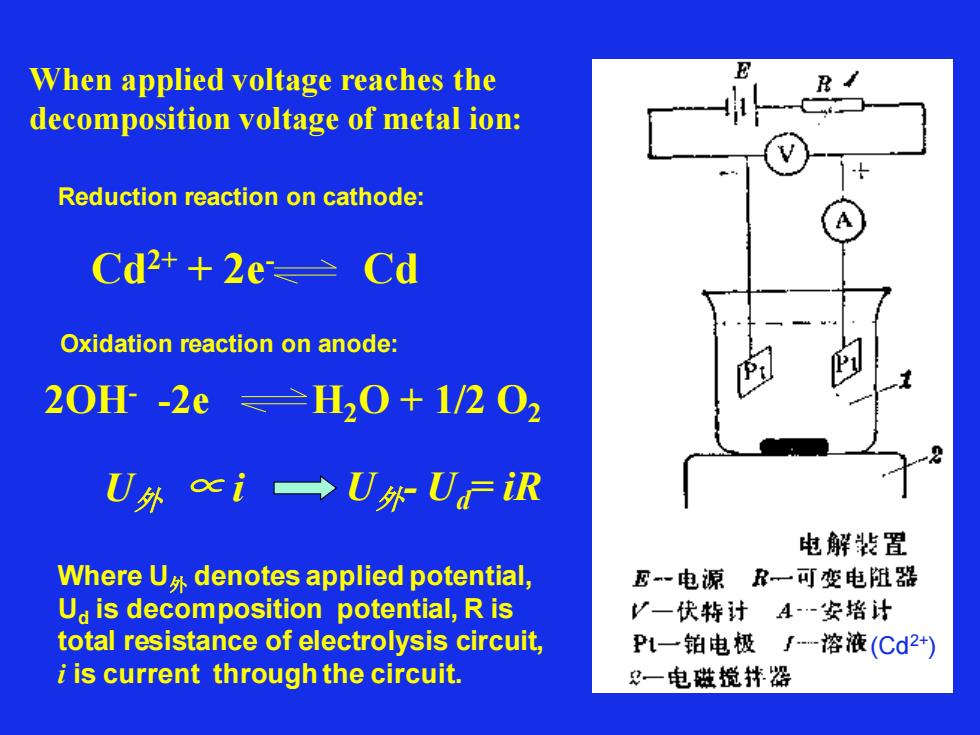
When applied voltage reaches the decomposition voltage of metal ion: Reduction reaction on cathode: Cd2+ + 2e- Cd Oxidation reaction on anode: 2OH- -2e H2O + 1/2 O2 U外 ∝ i Where U外 denotes applied potential, Ud is decomposition potential, R is total resistance of electrolysis circuit, i is current through the circuit. U外- Ud= iR (Cd2+)
When applied voltage reaches the decomposition voltage of metal ion: Reduction reaction on cathode: Cd2+ + 2e- Cd Oxidation reaction on anode: 2OH- -2e H2O + 1/2 O2 U外 ∝ i Where U外 denotes applied potential, Ud is decomposition potential, R is total resistance of electrolysis circuit, i is current through the circuit. U外- Ud= iR (Cd2+)
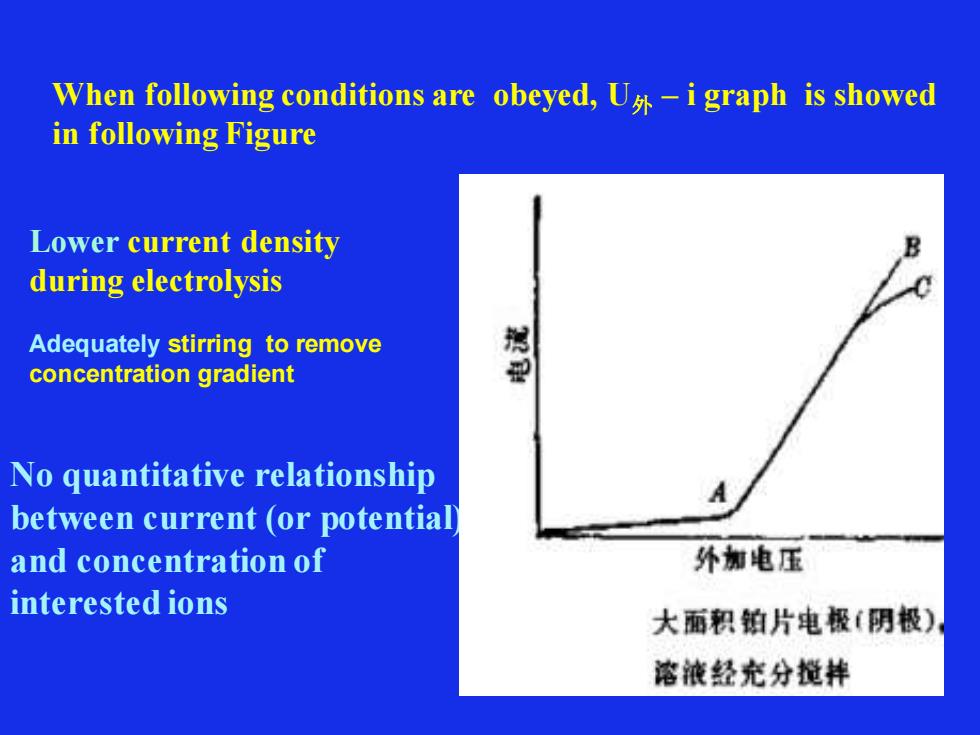
When following conditions are obeyed, U外 – i graph is showed in following Figure Lower current density during electrolysis Adequately stirring to remove concentration gradient No quantitative relationship between current (or potential) and concentration of interested ions
When following conditions are obeyed, U外 – i graph is showed in following Figure Lower current density during electrolysis Adequately stirring to remove concentration gradient No quantitative relationship between current (or potential) and concentration of interested ions

To find the analytical relationship between the current and the concentration of sought-for ions , following measurements are adopted: ●MICROPLATINUM ELECTRODE Or DROPPING MERCURY ELECTRODE ( in most common use) to insure high current density ● NO STIRRING to insure enough high concentration difference between the electrode surface and the solution bulk Then E = Eo + ln CM RT nF
To find the analytical relationship between the current and the concentration of sought-for ions , following measurements are adopted: ●MICROPLATINUM ELECTRODE Or DROPPING MERCURY ELECTRODE ( in most common use) to insure high current density ● NO STIRRING to insure enough high concentration difference between the electrode surface and the solution bulk Then E = Eo + ln CM RT nF